Wherever you are in your product development lifecycle,
Sterling can help.
P: 201-877-5682
F: 201-301-9169
info@sterlingmedicaldevices.com

We’ll help you navigate the complex regulations, processes and protocols to achieve regulatory compliance, on-schedule. Backed by proven procedures and tools, our team has extensive experience helping multinational companies with their EU MDR and IVDR transitions and implementations.
Our team of scientific writers will pull together and create the required documentation to ensure you comply with EU MDR, IVDR, and Medical Device Directive (MDD) requirements, including:
For more information on the EU MDR and IVDR regulatory processes, reach out to our team today.

July 28, 2021

February 18, 2021
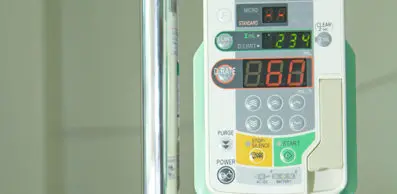
September 8, 2020